As I delved into the world of telomeres, I was captivated by the idea that these tiny DNA structures could hold secrets to aging and health. The sheer thought that the ends of our chromosomes might influence how we age was enough to make me rethink everything I knew about longevity.
What Are Telomeres and Their Role in Aging?
To understand the connection between telomeres and aging, it helps to start with what telomeres actually are. Telomeres are protective DNA sequences located at the ends of each chromosome. Humans have 23 pairs of chromosomes, and these telomere caps serve a vital function: they protect the chromosome ends from damage during cell division.
The Protective Role of Telomeres in Cell Division
Every time a cell divides, it must replicate its DNA so that both new cells receive a full set of genetic information. However, due to the way DNA replication works, the very ends of chromosomes cannot be fully copied. This means that with each round of cell division, telomeres become slightly shorter.
Think of telomeres as the plastic tips at the end of shoelaces—they prevent the chromosome “laces” from fraying or sticking to each other. Without these protective caps, important genetic material could be lost or damaged during replication.
How Telomere Shortening Occurs Over Time
This gradual shortening happens because DNA polymerase—the enzyme responsible for copying DNA—cannot completely replicate the very end segments of linear chromosomes. As a result:
- Telomeres act as buffers that absorb this loss without affecting essential genes.
- With every cell division, telomere length decreases incrementally.
- Eventually, when telomeres become too short, cells can no longer divide effectively and enter a state called cellular senescence or programmed death.
This process is natural but accumulates over time as our cells continue dividing throughout life.
The Link Between Telomere Length and Aging
The shortening of telomeres has been closely associated with biological aging. As cells lose their ability to divide due to critically short telomeres, tissue regeneration slows down and organ function may decline. This cellular aging contributes to many age-related changes in our bodies.
Scientific studies have found correlations between shorter telomere length and increased markers of aging as well as higher risks for certain diseases:
- A review from 2011 noted that signs of DNA damage and reduced telomere function tend to increase with age.
- A smaller study in 2003 linked shorter telomeres with greater mortality risk from heart disease and infectious diseases among participants.
- Larger meta-analyses suggest connections between shortened telomeres and coronary heart disease or some cancers; however, links to overall mortality remain less clear-cut.
The Role of Telomerase Enzyme
An important factor in this process is an enzyme called telomerase. This enzyme can add back DNA repeats onto shortened telomeres in certain types of cells—such as stem cells and germ cells—helping maintain their length over time. However:
- Most human somatic (body) cells do not produce enough telomerase activity to prevent gradual shortening during normal cell divisions.
- Cancer cells often reactivate high levels of telomerase which allows them to divide uncontrollably by maintaining their telomere length indefinitely—a hallmark feature seen in over 90% of cancers.
The Connection Between Telomeric Changes and Disease Risk
The relationship between shorter telomeres and disease risk is complex but significant:
- Aging-related diseases: Shortened telomeres may contribute to decreased tissue repair capacity leading to conditions like cardiovascular disease or weakened immune responses.
- Cancer: While short telomeres can trigger genomic instability promoting cancer development initially, cancerous tumors often overcome this by reactivating telomerase for unlimited growth potential.
Oxidative Stress: A Driver Behind Telomere Shortening?
An additional factor influencing how quickly our telomeres shorten is oxidative stress—the damage caused by reactive oxygen species (ROS). These molecules arise naturally through metabolism but also increase due to inflammation or environmental exposures such as pollution, smoking, or alcohol consumption. Oxidative stress damages DNA including at chromosome ends which may accelerate shortening beyond normal rates associated solely with replication cycles.
A Summary on Telomers’ Role in Aging Biology
| Key Point | Description |
|---|---|
| Telomers Definition | Dna sequences protecting chromosome ends during cell division |
| Teloermer Shortening Process | Slightly shorter after every cell replication cycle |
| Aging Connection | Tied to cellular senescence & increased risk for age-related diseases |
| Telomerase Enzyme | Adds repeats back onto some cell types' teloermes; low activity elsewhere |
| Disease Implications | Cancer uses reactivated teloermase; oxidative stress accelerates shortening |
I find it fascinating how these tiny structures at chromosome tips play such an outsized role in our biology—acting as both guardians against genetic loss during cell division and markers signaling when our bodies begin showing signs of aging at a cellular level. Understanding how they work gives us insight into fundamental processes behind healthspan and longevity research today.
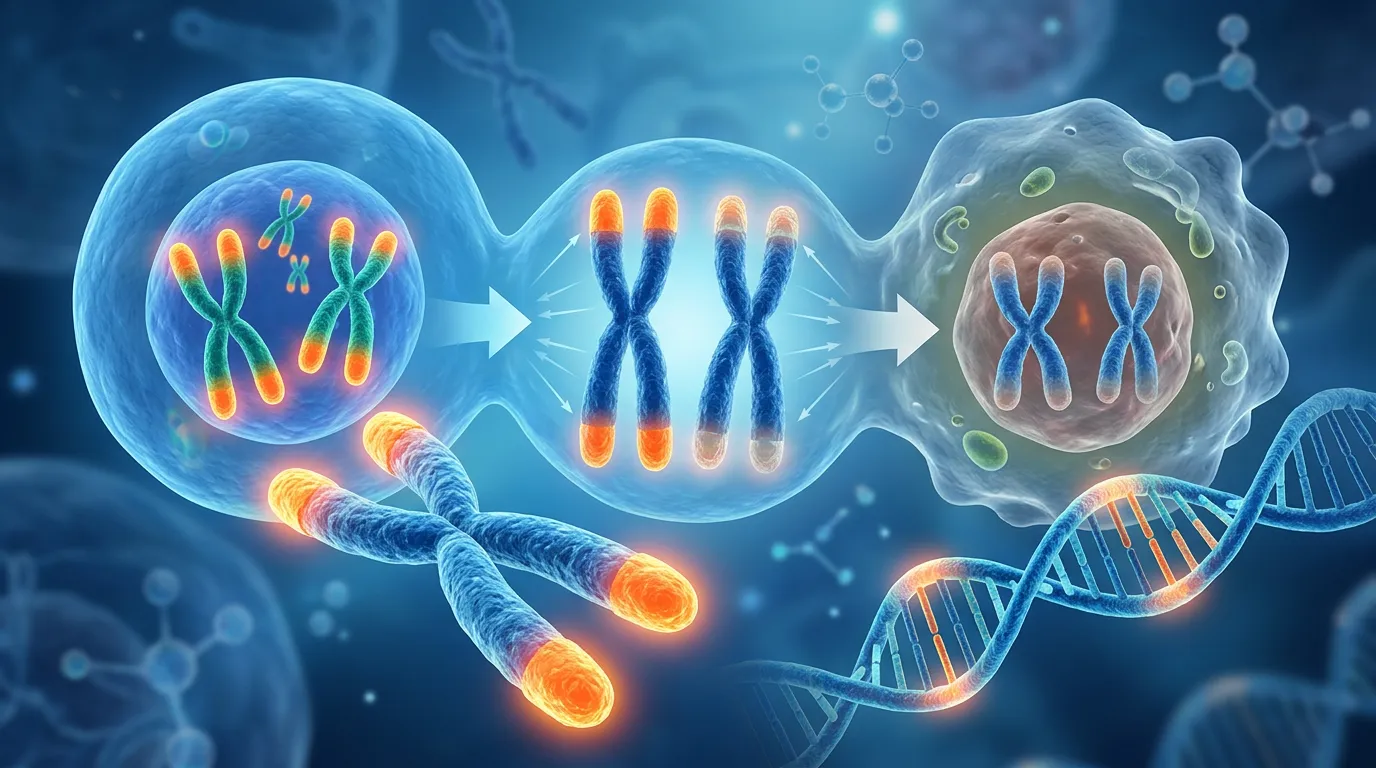
The Connection Between Telomere Length and Lifestyle Factors
Understanding how lifestyle choices affect telomere length is a fascinating area of research that bridges genetics, aging, and overall health. Telomeres, the protective caps at the ends of our chromosomes, naturally shorten as cells divide. This shortening process is linked to aging and various diseases. While we cannot stop this process entirely, emerging evidence suggests that certain lifestyle factors might influence the rate of telomere shortening or even promote telomere lengthening in some cases.
Healthy Lifestyle and Telomere Lengthening: What Does the Research Say?
One key question many people ask is whether adopting a healthy lifestyle can actually lengthen telomeres or slow their shortening. The current scientific understanding indicates that while outright lengthening of telomeres remains rare outside specialized cells with active telomerase enzyme, slowing down their shortening is more achievable through lifestyle changes.
A small but intriguing 2013 pilot study involving men with low-risk prostate cancer found that those who adopted comprehensive healthy habits—including improved diet, regular exercise, and stress management—showed longer telomeres five years later compared to those who did not make such changes. Although this study was limited in size and scope, it highlights potential benefits worth exploring further.
The Impact of Diet on Telomere Health
Diet plays an important role in maintaining cellular health and may influence telomere dynamics through its effects on inflammation and oxidative stress. For example:
- Mediterranean-style diets, rich in antioxidants from fruits, vegetables, nuts, whole grains, and healthy fats like olive oil, have been associated with better telomere integrity.
- A large 2018 study involving over 5,000 adults found a positive link between higher dietary fiber intake and longer telomeres. Fiber’s ability to help regulate blood sugar levels and reduce inflammation could be key mechanisms behind this association.
- However, other studies suggest that diet alone may not fully determine telomere length; genetics and other non-dietary factors also contribute significantly.
Overall, focusing on nutrient-rich foods that combat oxidative stress seems beneficial for preserving telomeres as part of a balanced diet.
Exercise: A Strong Ally for Telomere Maintenance
Among lifestyle factors studied so far, physical activity shows one of the most consistent associations with longer telomeres. Several studies highlight this connection:
- A comprehensive 2017 US study involving thousands of participants demonstrated that individuals engaging in high levels of physical activity had significantly longer telomeres than those with low or medium activity levels.
- A separate study from the same year focused on young adults found links between aerobic fitness combined with muscle endurance exercises and increased telomere length.
This evidence suggests regular aerobic exercise—such as walking briskly, cycling, swimming—and strength training can support cellular health by potentially slowing down the natural erosion of chromosome ends.
Practical Tip:
- If you’re looking to enhance your workout routine for better cellular health outcomes, incorporating at least 10 minutes daily of moderate aerobic exercises can be a good start.
The Role of Stress Management in Protecting Telomeres
Chronic psychological stress has been shown to accelerate biological aging partly through its impact on oxidative stress levels within cells. Stress hormones like cortisol increase oxidative damage to DNA—including at the vulnerable ends where telomeres reside—which can speed up their shortening process.
"Studies show chronic caregiving stress correlates strongly with shorter telomeres," emphasizing how emotional wellbeing ties into cellular longevity.
This connection underscores why managing stress through mindfulness practices, meditation, adequate sleep quality, or therapy could be valuable strategies for maintaining healthier telomeres over time.
The Link Between Oxidative Stress and Telomere Shortening
Oxidative stress, caused by reactive oxygen species (ROS) generated during normal metabolism as well as exposure to environmental toxins like pollution or smoking, damages DNA molecules including those within our chromosomes’ protective caps—the telomeres. This damage accelerates their degradation beyond what occurs during normal cell division alone.
| Factor Influencing Oxidative Stress | Description |
|---|---|
| Environmental Toxins (e.g., pollution) | Create excess ROS leading to DNA damage including shortened telomeres |
| Lifestyle Habits (e.g., smoking & alcohol) | Add oxidative burden accelerating cellular aging processes |
| Diet Rich in Antioxidants (e.g., Mediterranean diet) | Counters ROS effects helping protect DNA integrity including at chromosome ends |
| Physical Activity & Stress Management | Lowers systemic inflammation reducing oxidative damage risk |
Tackling oxidative stress through antioxidant-rich diets combined with regular exercise and effective stress reduction techniques forms a promising approach toward supporting healthier aging via preservation of our genetic material’s stability—including maintaining optimal telomere length.

Can We Truly Lengthen Our Telomeres?
The question of whether we can actively lengthen our telomeres is both fascinating and complex. Telomeres, the protective caps at the ends of chromosomes, naturally shorten as cells divide, a process linked to aging and various diseases. While the enzyme telomerase has the ability to extend telomeres in certain cell types, most human cells do not produce enough telomerase to prevent this shortening over time. This biological reality sets the stage for ongoing scientific debate about whether it is possible—and safe—to modify telomere length meaningfully in humans.
From a scientific standpoint, much of the current research focuses on slowing down telomere shortening rather than reversing it entirely. Early studies have provided some intriguing insights but stop short of offering definitive answers. For example, a small 2013 pilot study involving ten men with low-risk prostate cancer found that those who adopted comprehensive lifestyle changes—including diet improvements, regular exercise, and stress management—showed longer telomeres after five years compared to a control group that did not make these changes. Although promising, this study’s limited size means its findings should be interpreted cautiously.
These lifestyle interventions are thought to influence telomere dynamics by reducing oxidative stress and inflammation—two factors known to accelerate telomere shortening. Oxidative stress results from reactive oxygen species produced during normal metabolism and heightened by environmental exposures such as pollution or smoking. By adopting antioxidant-rich diets like the Mediterranean diet or increasing fiber intake through whole foods, individuals may help protect their DNA from damage that contributes to faster telomere erosion.
Exercise also appears consistently linked with healthier telomere profiles. Research involving thousands of participants has shown that higher levels of physical activity correlate with longer telomeres compared to sedentary lifestyles. Aerobic fitness and muscle endurance specifically seem beneficial for maintaining chromosome integrity over time.
Stress management plays an equally important role because chronic psychological stress elevates cortisol levels and oxidative damage, which can accelerate cellular aging processes including telomere attrition. Studies have demonstrated associations between caregiving stress or heightened cortisol responses and shorter telomeres, reinforcing how emotional well-being intersects with molecular health.
The role of telomerase itself adds another layer of complexity. While most normal human cells lack significant amounts of this enzyme, many cancer cells reactivate it—allowing them to maintain their telomeres indefinitely and proliferate uncontrollably. This discovery has spurred interest in developing therapies targeting telomerase activity as potential cancer treatments but also raises caution about artificially boosting this enzyme in healthy tissues due to potential risks like increased cancer susceptibility.
Given these nuances, the feasibility of intentional telomere lengthening remains uncertain at present. The emerging consensus among researchers emphasizes prevention: adopting healthy lifestyle habits that may slow down natural shortening rather than attempting direct elongation through experimental interventions or supplements whose safety profiles are not fully established.
This balanced view aligns with broader health advice advocating for diets rich in antioxidants and fiber, regular physical activity tailored to individual capacity, effective stress reduction techniques such as mindfulness or therapy, and avoidance of harmful exposures like smoking or excessive alcohol consumption—all contributing factors potentially supportive of better cellular health including more stable telomeres.
In summary, while we cannot yet claim with certainty that we can truly lengthen our telomeres on demand, there is growing evidence suggesting that lifestyle choices can influence how quickly they shorten—a subtle but meaningful form of biological preservation. As research advances into understanding mechanisms behind telomerase, oxidative stress modulation, and genetic influences on aging pathways, future therapies might one day offer more direct methods for modifying chromosome end protection safely.
Until then, focusing on holistic wellness strategies remains our best approach—not only for supporting healthy aging at the molecular level but also for improving overall quality of life across physical and mental dimensions.
TL;DR: Telomeres are crucial to aging; while some lifestyle changes may help, the science of lengthening them is not definitive yet.

Comments
Post a Comment